| Citation: | Yu W, Yao N, Pan J, Fang W, Li X et al. Highly sensitive and fast response strain sensor based on evanescently coupled micro/nanofibers. Opto-Electron Adv 5, 210101 (2022). doi: 10.29026/oea.2022.210101 |
Highly sensitive and fast response strain sensor based on evanescently coupled micro/nanofibers
-
Abstract
Flexible strain sensors play an important role in electronic skins, wearable medical devices, and advanced robots. Herein, a highly sensitive and fast response optical strain sensor with two evanescently coupled optical micro/nanofibers (MNFs) embedded in a polydimethylsiloxane (PDMS) film is proposed. The strain sensor exhibits a gauge factor as high as 64.5 for strain ≤ 0.5% and a strain resolution of 0.0012% which corresponds to elongation of 120 nm on a 1 cm long device. As a proof-of-concept, highly sensitive fingertip pulse measurement is realized. The properties of fast temporal frequency response up to 30 kHz and a pressure sensitivity of 102 kPa−1 enable the sensor for sound detection. Such versatile sensor could be of great use in physiological signal monitoring, voice recognition and micro-displacement detection.-
Keywords:
- optical micro/nanofiber /
- strain sensor /
- pressure sensor /
- micro-displacement /
- directional coupler
-
-
References
[1] Wang XD, Dong L, Zhang HL, Yu RM, Pan CF et al. Recent progress in electronic skin. Adv Sci 2, 1500169 (2015). doi: 10.1002/advs.201500169 [2] Yang JC, Mun J, Kwon SY, Park S, Bao ZN et al. Electronic skin: recent progress and future prospects for skin-attachable devices for health monitoring, robotics, and prosthetics. Adv Mater 31, 1904765 (2019). doi: 10.1002/adma.201904765 [3] Someya T, Bao ZN, Malliaras G G. The rise of plastic bioelectronics. Nature 540, 379–385 (2016). doi: 10.1038/nature21004 [4] Chen Y, Zhang YC, Liang ZW, Cao Y, Han ZY et al. Flexible inorganic bioelectronics. npj Flex Electron 4, 2 (2020). doi: 10.1038/s41528-020-0065-1 [5] Takei K, Honda W, Harada S, Arie T, Akita S. Toward flexible and wearable human-interactive health-monitoring devices. Adv Healthc Mater 4, 487–500 (2015). doi: 10.1002/adhm.201400546 [6] Liu YH, Pharr M, Salvatore GA. Lab-on-skin: a review of flexible and stretchable electronics for wearable health monitoring. ACS Nano 11, 9614–9635 (2017). doi: 10.1021/acsnano.7b04898 [7] Tan FZ, Lyu WM, Chen SY, Liu ZY, Yu CY. Contactless vital signs monitoring based on few-mode and multi-core fibers. Opto-Electron Adv 3, 190034 (2020). doi: 10.29026/oea.2020.190034 [8] Whitesides GM. Soft robotics. Angew Chem Int Ed 57, 4258–4273 (2018). doi: 10.1002/anie.201800907 [9] Van Meerbeek IM, De Sa CM, Shepherd RF. Soft optoelectronic sensory foams with proprioception. Sci Robot 3, eaau2489 (2018). doi: 10.1126/scirobotics.aau2489 [10] Zhang SM, Cai L, Li W, Miao JS, Wang TY et al. Fully printed silver-nanoparticle-based strain gauges with record high sensitivity. Adv Electron Mater 3, 1700067 (2017). doi: 10.1002/aelm.201700067 [11] Li LH, Xiang HY, Xiong Y, Zhao H, Bai YY et al. Ultrastretchable fiber sensor with high sensitivity in whole workable range for wearable electronics and implantable medicine. Adv Sci 5, 1800558 (2018). doi: 10.1002/advs.201800558 [12] Wang B, Wu K, Hjort K, Guo CF, Wu ZG. High-performance liquid alloy patterning of epidermal strain sensors for local fine skin movement monitoring. Soft Robot 6, 414–421 (2019). doi: 10.1089/soro.2018.0008 [13] Chen DJ, Zhao XL, Wei XR, Zhang JL, Wang D et al. Ultrastretchable, tough, antifreezing, and conductive cellulose hydrogel for wearable strain sensor. ACS Appl Mater Interfaces 12, 53247–53256 (2020). doi: 10.1021/acsami.0c14935 [14] Luo ZW, Li XM, Li QL, Tian XY, Fan TY et al. In situ dynamic manipulation of graphene strain sensor with drastically sensing performance enhancement. Adv Electron Mater 6, 2000269 (2020). doi: 10.1002/aelm.202000269 [15] Lin WE, He CB, Huang HY, Zhao WY, Qiu YB et al. Simultaneously achieving ultrahigh sensitivity and wide detection range for stretchable strain sensors with an interface-locking strategy. Adv Mater Technol 5, 2000008 (2020). doi: 10.1002/admt.202000008 [16] Wang BZ, Ba DX, Chu Q, Qiu LQ, Zhou DW et al. High-sensitivity distributed dynamic strain sensing by combining Rayleigh and Brillouin scattering. Opto-Electron Adv 3, 200013 (2020). doi: 10.29026/oea.2020.200013 [17] Wang S, Fang YL, He H, Zhang L, Li CA et al. Wearable stretchable dry and self-adhesive strain sensors with conformal contact to skin for high-quality motion monitoring. Adv Funct Mater 31, 2007495 (2021). doi: 10.1002/adfm.202007495 [18] Liu ZY, Qi DP, Guo PZ, Liu Y, Zhu BW et al. Thickness-gradient films for high gauge factor stretchable strain sensors. Adv Mater 27, 6230–6237 (2015). doi: 10.1002/adma.201503288 [19] Yamada T, Hayamizu Y, Yamamoto Y, Yomogida Y, Izadi-Najafabadi A et al. A stretchable carbon nanotube strain sensor for human-motion detection. Nat Nanotechnol 6, 296–301 (2011). doi: 10.1038/nnano.2011.36 [20] Ke K, Pötschke P, Wiegand N, Krause B, Voit B. Tuning the network structure in poly(vinylidene fluoride)/carbon nanotube nanocomposites using carbon black: toward improvements of conductivity and piezoresistive sensitivity. ACS Appl Mater Interfaces 8, 14190–14199 (2016). doi: 10.1021/acsami.6b03451 [21] Kang D, Pikhitsa PV, Choi YW, Lee C, Shin SS et al. Ultrasensitive mechanical crack-based sensor inspired by the spider sensory system. Nature 516, 222–226 (2014). doi: 10.1038/nature14002 [22] Chen S, Wei Y, Wei SM, Lin Y, Liu L. Ultrasensitive Cracking-assisted strain sensors based on silver nanowires/graphene hybrid particles. ACS Appl Mater Interfaces 8, 25563–25570 (2016). doi: 10.1021/acsami.6b09188 [23] Amjadi M, Turan M, Clementson CP, Sitti M. Parallel microcracks-based ultrasensitive and highly stretchable strain sensors. ACS Appl Mater Interfaces 8, 5618–5626 (2016). doi: 10.1021/acsami.5b12588 [24] Song HL, Zhang JQ, Chen DB, Wang KJ, Niu SC et al. Superfast and high-sensitivity printable strain sensors with bioinspired micron-scale cracks. Nanoscale 9, 1166–1173 (2017). doi: 10.1039/C6NR07333F [25] Ye JL, Yang TT, Zhang YF, Lin L. Single-crack-activated ultrasensitive impedance strain sensor. Adv Mater Interfaces 5, 1800616 (2018). doi: 10.1002/admi.201800616 [26] Guo X, Ying YB, Tong LM. Photonic nanowires: from subwavelength waveguides to optical sensors. Acc Chem Res 47, 656–666 (2014). doi: 10.1021/ar400232h [27] Zhang L, Tang Y, Tong LM. Micro-/nanofiber optics: merging photonics and material science on nanoscale for advanced sensing technology. iScience 23, 100810 (2020). doi: 10.1016/j.isci.2019.100810 [28] Zhang L, Pan J, Zhang Z, Wu H, Yao N et al. Ultrasensitive skin-like wearable optical sensors based on glass micro/nanofibers. Opto-Electron Adv 3, 190022 (2020). [29] Pan J, Zhang Z, Jiang CP, Zhang L, Tong LM. A multifunctional skin-like wearable optical sensor based on an optical micro-/nanofibre. Nanoscale 12, 17538–17544 (2020). doi: 10.1039/D0NR03446K [30] Tang Y, Liu HT, Pan J, Zhang Z, Xu Y et al. Optical micro/nanofiber-enabled compact tactile sensor for hardness discrimination. ACS Appl Mater Interfaces 13, 4560–4566 (2021). doi: 10.1021/acsami.0c20392 [31] Huang KJ, Yang SY, Tong LM. Modeling of evanescent coupling between two parallel optical nanowires. Appl Opt 46, 1429–1434 (2007). doi: 10.1364/AO.46.001429 [32] Chen Y, Yan SC, Zheng X, Xu F, Lu YQ. A miniature reflective micro-force sensor based on a microfiber coupler. Opt Express 22, 2443–2450 (2014). doi: 10.1364/OE.22.002443 [33] Liu TR, Pagliano F, van Veldhoven R, Pogoretskiy V, Jiao YQ et al. Integrated nano-optomechanical displacement sensor with ultrawide optical bandwidth. Nat Commun 11, 2407 (2020). doi: 10.1038/s41467-020-16269-7 [34] Chen Y, Ma Z, Yang Q, Tong LM. Compact optical short-pass filters based on microfibers. Opt Lett 33, 2565–2567 (2008). doi: 10.1364/OL.33.002565 [35] Lou Z, Chen S, Wang LL, Jiang K, Shen GZ. An ultra-sensitive and rapid response speed graphene pressure sensors for electronic skin and health monitoring. Nano Energy 23, 7–14 (2016). doi: 10.1016/j.nanoen.2016.02.053 [36] Li SM, Xiao XL, Hu JY, Dong MC, Zhang YQ et al. Recent advances of carbon-based flexible strain sensors in physiological signal monitoring. ACS Appl Electron Mater 2, 2282–2300 (2020). doi: 10.1021/acsaelm.0c00292 [37] Fan XY, Huang Y, Ding XR, Luo NQ, Li CL et al. Alignment-free liquid-capsule pressure sensor for cardiovascular monitoring. Adv Funct Mater 28, 1805045 (2018). doi: 10.1002/adfm.201805045 [38] Lin QP, Huang J, Yang JL, Huang Y, Zhang YF et al. Highly sensitive flexible iontronic pressure sensor for fingertip pulse monitoring. Adv Healthc Mater 9, 2001023 (2020). doi: 10.1002/adhm.202001023 [39] Wang CY, Li X, Gao EL, Jian MQ, Xia KL et al. Carbonized silk fabric for ultrastretchable, highly sensitive, and wearable strain sensors. Adv Mater 28, 6640–6648 (2016). doi: 10.1002/adma.201601572 [40] Gong S, Lai DTH, Su B, Si KJ, Ma Z et al. Highly stretchy black gold E-skin nanopatches as highly sensitive wearable biomedical sensors. Adv Electron Mater 1, 1400063 (2015). doi: 10.1002/aelm.201400063 [41] Nichols WW. Clinical measurement of arterial stiffness obtained from noninvasive pressure waveforms. Am J Hypertens 18, 3S–10S (2005). [42] Choong CL, Shim MB, Lee BS, Jeon S, Ko DS et al. Highly stretchable resistive pressure sensors using a conductive elastomeric composite on a micropyramid array. Adv Mater 26, 3451–3458 (2014). doi: 10.1002/adma.201305182 [43] Yang LY, Li YP, Fang F, Li LY, Yan ZJ et al. Highly sensitive and miniature microfiber-based ultrasound sensor for photoacoustic tomography. Opto-Electron Adv 4, 200076 (2022). -
Supplementary Information
Supplymentray information for Highly sensitive and fast response strain sensor based on evanescently coupled micro/nanofibers 
-
Access History
Article Metrics
-
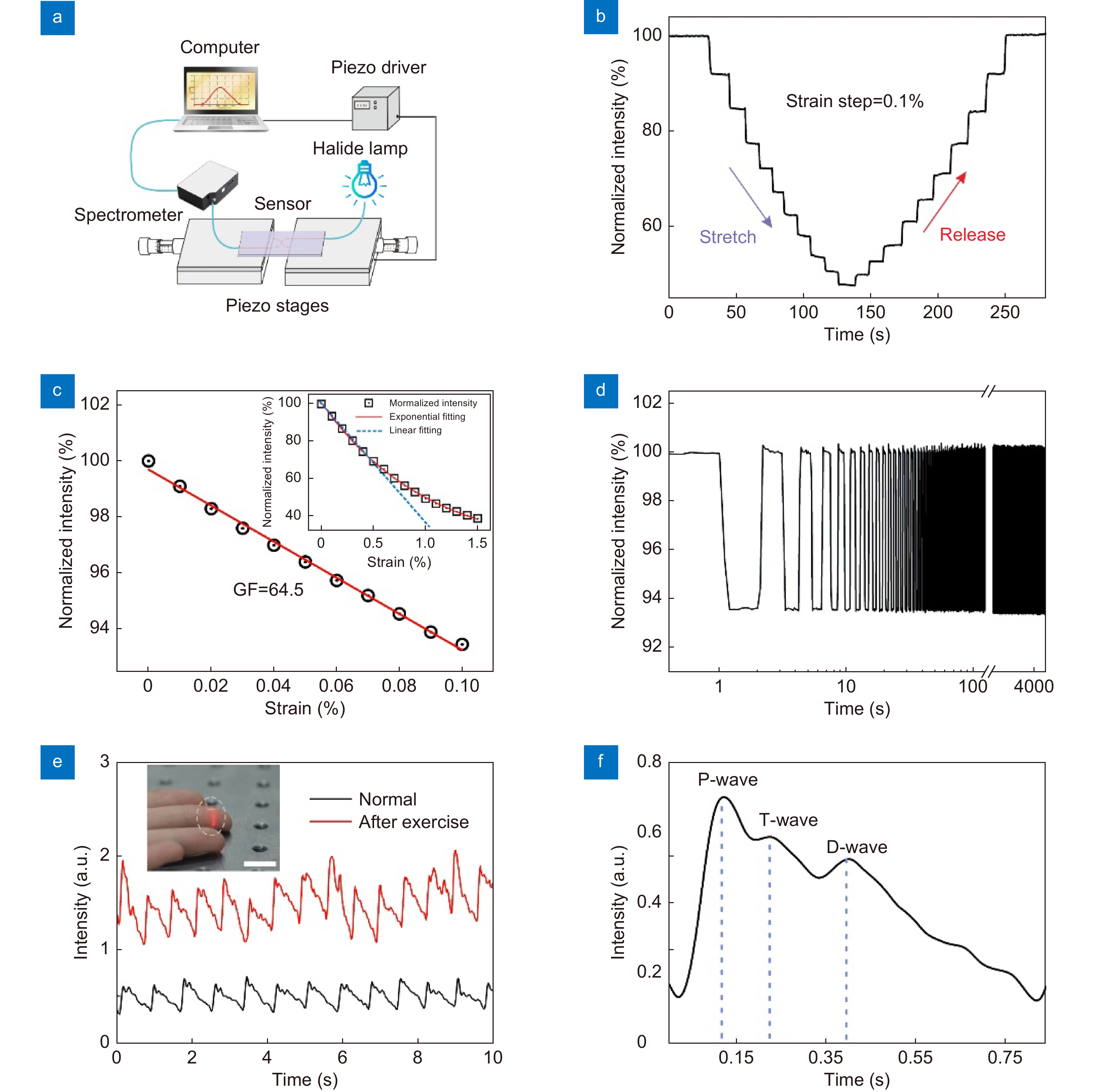
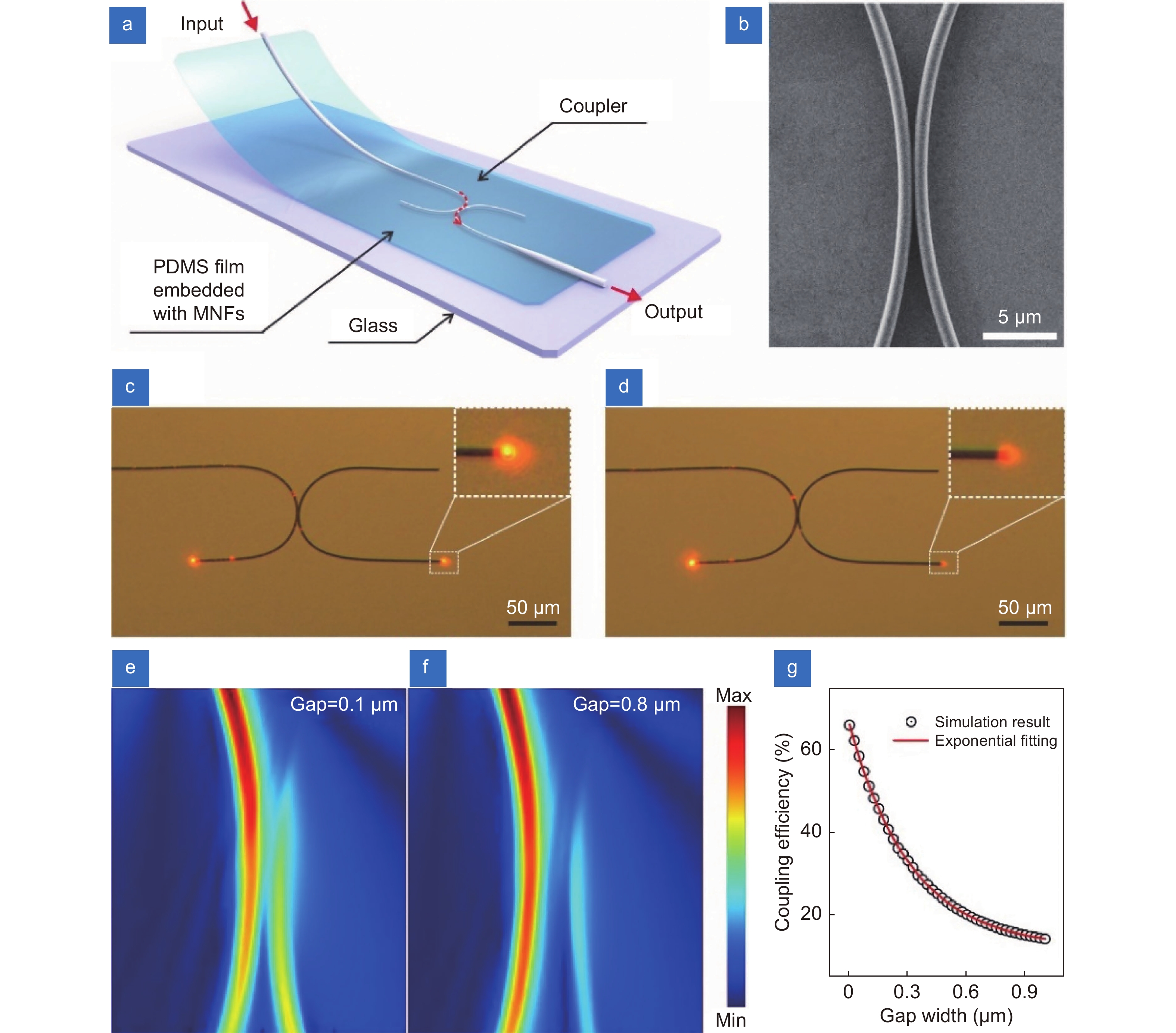
Figure 1.
Working principle of the strain sensor based on MNF coupler. (a) Schematic diagram of a strain sensor structure. (b) SEM image of the coupling region in a typical MNF coupler. (c, d) Optical micrographs of an MNF coupler before and after slightly changing of the gap, respectively. The wavelength of input light is 660 nm. (e, f) The simulated electromagnetic field intensity distributions of MNF coupling structures with gap widths of 0.1 μm and 0.8 μm, respectively. The diameter of each MNF is 0.9 μm and the bending radius is 50 μm. The refractive index of surrounding material is set as 1.40. (g) The coupling efficiency as a function of gap width of coupled MNFs.
-
Figure 2.
The strain response of the sensor. (a) Schematic diagram of the experimental setup for strain sensing. (b) The response of the sensor in a stretch and release cycle reveals good reversibility of the device. (c) Linear fitting of experimental data result in a GF of 64.5 within the strain range of 0.1%. Such linear relation can be extended to a strain range up to 0.5%, shown as the blue dotted-line in the inset, while the general relation between the normalized intensity and strain can be fitted by an exponential function in the strain range of 0–1.5%. (d) The durability test for 2000 cycles under 0.1% strain. (e) Measurement of the real-time fingertip pulse wave under normal conditions (72 beats per minute) and after exercise (85 beats per minute). The inset shows a photo of the sensor attached to the fingertip to test the pulse of the fingertip, and the scale bar is 1 cm. (f) Typical measured pulse waveform with distinguishable P-wave, T-wave and D-wave peaks.
-
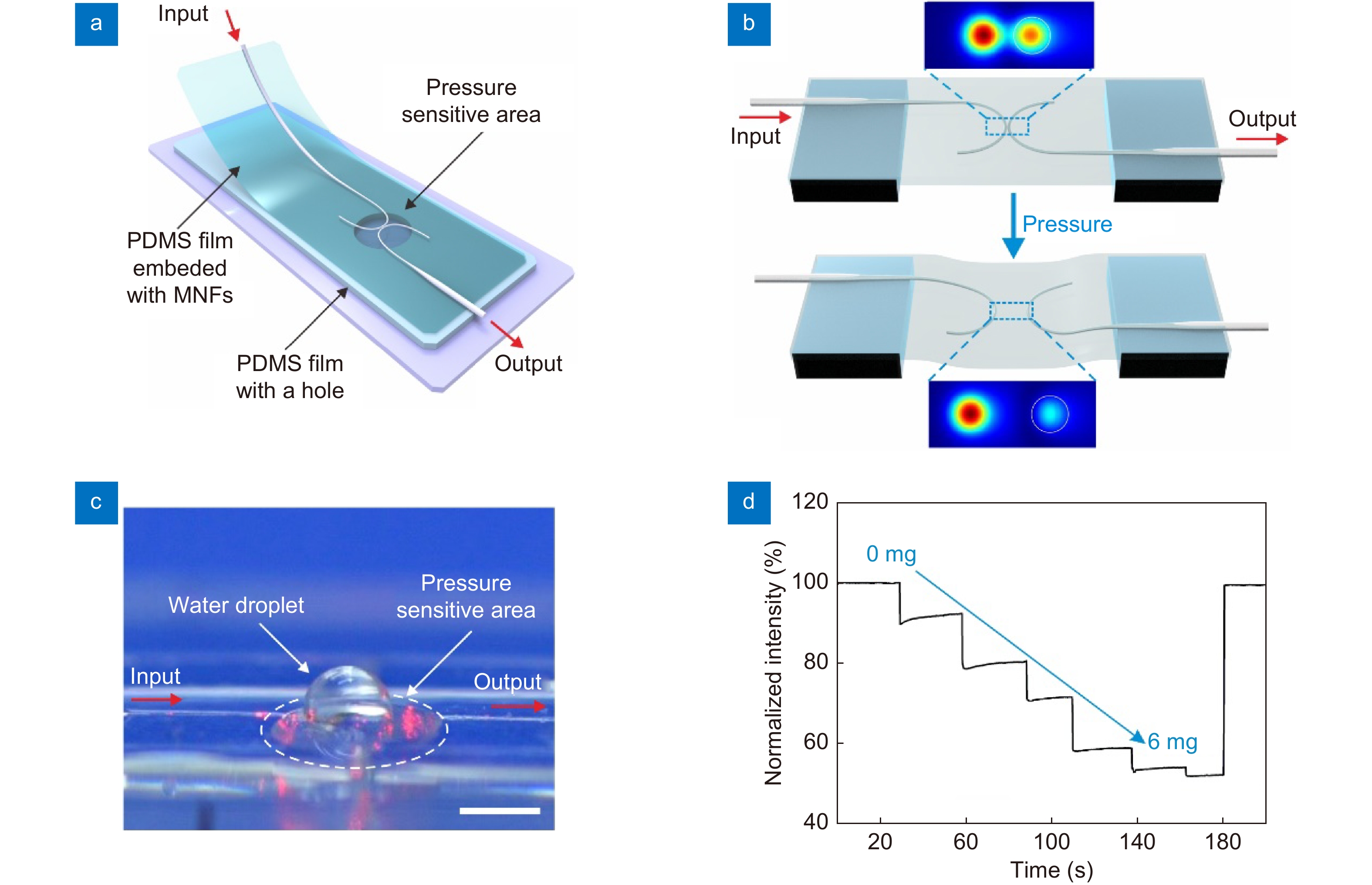
Figure 3.
Weak pressure signal detection. (a) Schematic diagram of the sensor structure. (b) Schematic diagram illustrates the pressure sensing mechanism. The vertical deformation of the film due to the exerted pressure causes the elongation of film. The enlargement of gap width results in a drop of coupling efficiency, as the simulated field distributions shown in the inset. The white circles indicate the location of MNFs. (c) A photo of the sensor with a water droplet on the pressure sensitive area, the scale bar is 1.5 mm. (d) The responses of the pressure sensor to water droplets with a maximum applied pressure of approximately 9 Pa.
-
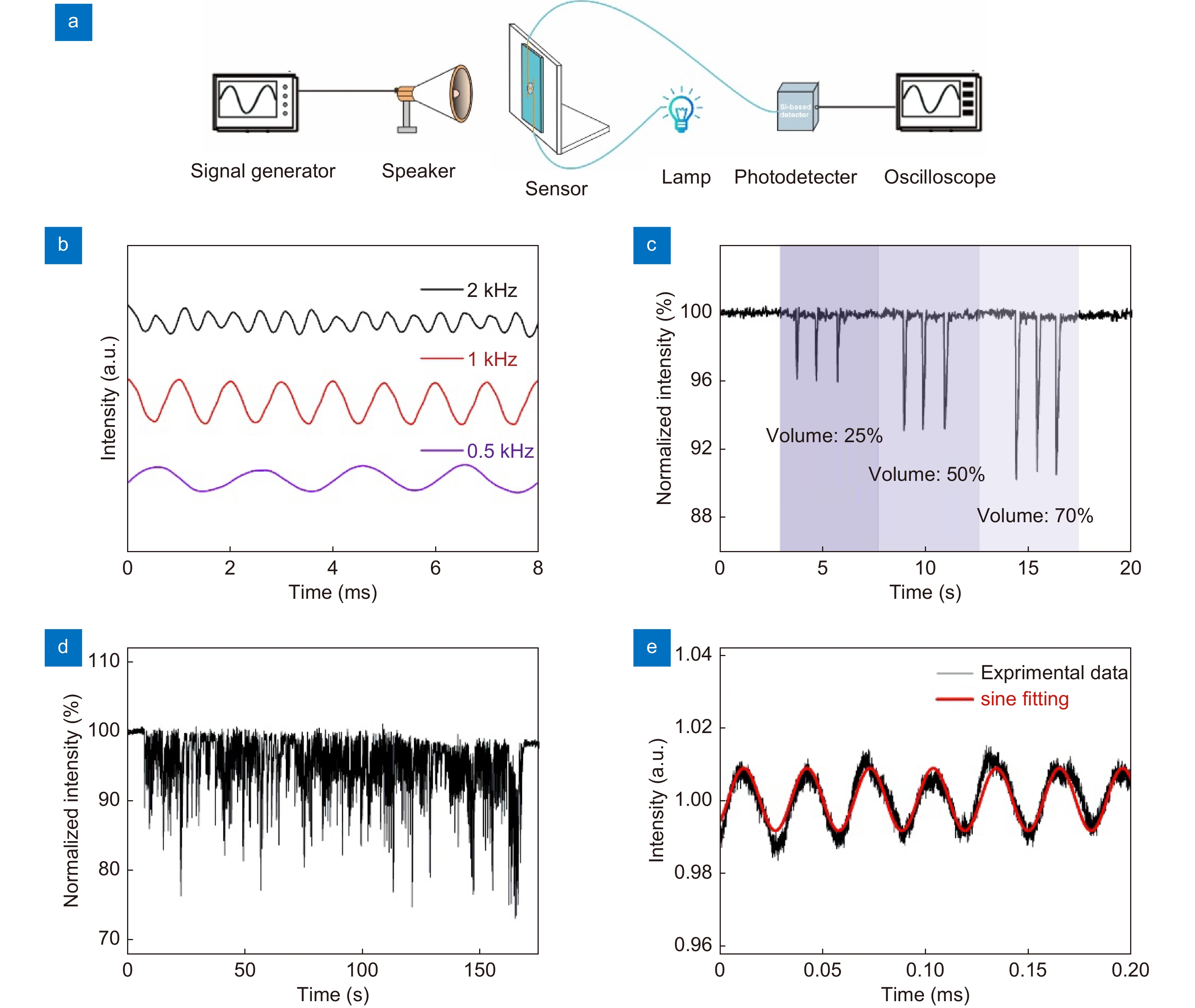
Figure 4.
Characterization of the sensor for sound detection. (a) Schematic diagram of the experimental setup for sound detection. (b) The device response to sinusoidal sound waves at 0.5 kHz, 1 kHz, 2 kHz, respectively. (c) The device response to a metronome at three different volumes (25, 50, and 70%). (d) The device response to a piece of music. (e) The sensor’s response to ultrasonic vibrations with a frequency of 30 kHz.

 E-mail Alert
E-mail Alert RSS
RSS

 DownLoad:
DownLoad: